DPRA
The DPRA (Direct Peptide Reactivity Assay) is a in Chemico evaluation of the sensitizing potency by measuring the reactivity of a compound with a reference peptide.
OECD 442C
Cover the key event 1 (Molecular events)
H-CLAT
The h-CLAT is a method for skin sensitization by quantifying changes in the expression of cell surface markers associated with the process of activation of Denditric Cells (CD86 and CD54), in the human leukemia cell line THP-1.
Following exposure to sensitisers, the measured expression levels of CD86 and CD54 cell surface markers are then used for supporting the discrimination between skin sensitisers and non-sensitisers.
OECD 442E
Cover Key event 3 (Activation of dendritic cells)
SENS-IS
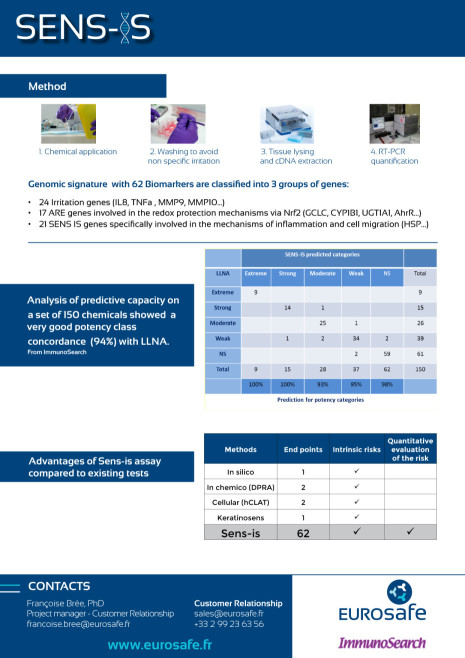
The Sens-IS assay, developped by Immunosearch, measures quantitatively the over expression of 62 specific biomarkers of skin sensitization, irritation and Redox system by RT-PCR after application of chemicals, mixture, natural extracts on 3D reconstructed human epidermis.
- Qualitative and quantitative response
- Distinguishes irritation and sensitization
- 3D reconstitued epidermis = closer to the skin therefore a complete biological system accounting for multiple cellular responses
- Larger domain of applications than the other sensitization assays, with capacity to test lipophilic compounds
- Good compliance with LLNA
- Suitable for finished products
OECD validation is in progress.
Cover Key event 2 (Activation of keratinocytes)
Retrieve information about Sens-IS.

__GARD™
Eurosafe is official distributor of the GARD™in vitro assay (SenzaGen) for measuring Skin sensitization and Potency CLP/GHS classification based on dendritic cell lines. The test is a high accuracy genomic test measuring changes in gene expression of 200 genes relevant to the skin sensitization adverse outcome pathways (AOP).
OECD validation is in progress.
To have more technical information, please consult SenzaGen website.
Retrieve information about GARD™ Skin.


Eurosafe is also an official distributor of the GARD™ respiratory assay for respiratory sensitization.


